
The characteristics of cathode rays do not depend upon the material of electrodes or on the nature of gas present in cathode ray tube.This suggests that cathode rays consist of negatively charged particles, viz. When either electrical or magnetic field is present, cathode rays behave as negatively charged particles.When there is no electrical or magnetic field, these rays travel in straight lines.These rays are not visible but their behavior can be observed with the help of fluorescent materials because such materials glow when hit by cathode rays.Cathode rays move from cathode to anode.When these rays (after passing through anode) hit the coating, a bright spot is formed on the coating. 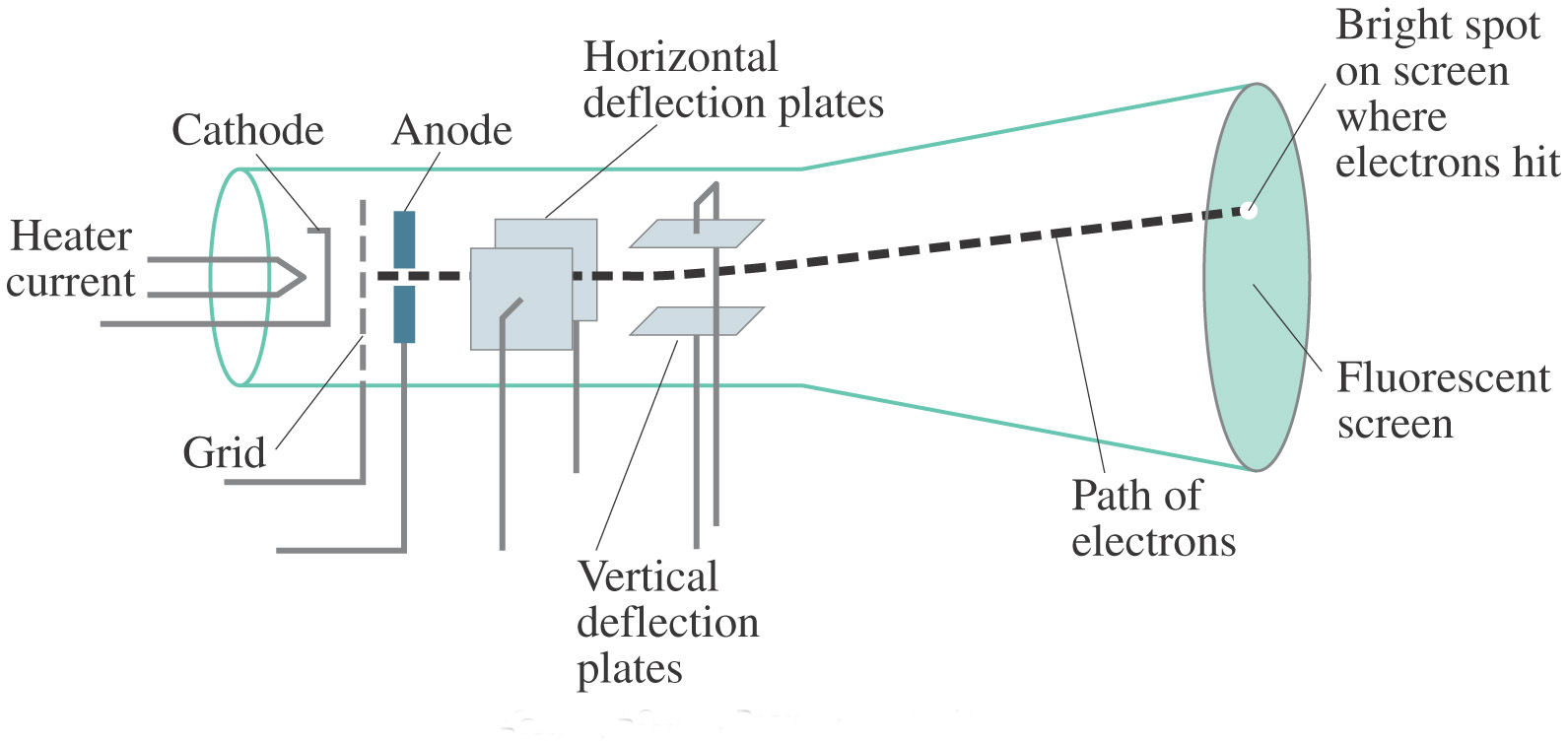
The flow of current was further checked by making a hole in the anode, and coating the tube (behind anode) with phosphorescent material zinc sulphide. These particles are called cathode rays or cathode ray particles. These particles move from the negative electrode (cathode) to the positive electrode (anode). When enough high voltage is applied across the electrodes, current starts to flow through a stream of particles. Evacuation of glass tube helps in manipulating the pressure of different gases in the tube. The electrical discharge through the gases can only be observed at very low pressure and at very high voltage. It contains two thin pieces of metal which are called electrodes, sealed inside. Structure of Cathode Ray Tube: This tube is made of glass. Faraday was the main scientist to conduct such experiments.īasic Concept Atomic Structure Periodic Table Chemical Bonding States of Matter Thermodynamics Equilibrium In mid 1850s many scientists began to study electrical discharge in cathode ray discharge tubes. These results suggested the particulate nature of electricity. The reaction result in liberation and deposition of matter at the electrodes. In 1830, Michael Faraday demonstrated that if electricity is passed through a solution of an electrolyte, it results in chemical reactions at electrodes. Structure of Atom Discovery of Subatomic Particles Discovery of Electron
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
